Our Science: The Cytocentric® Approach
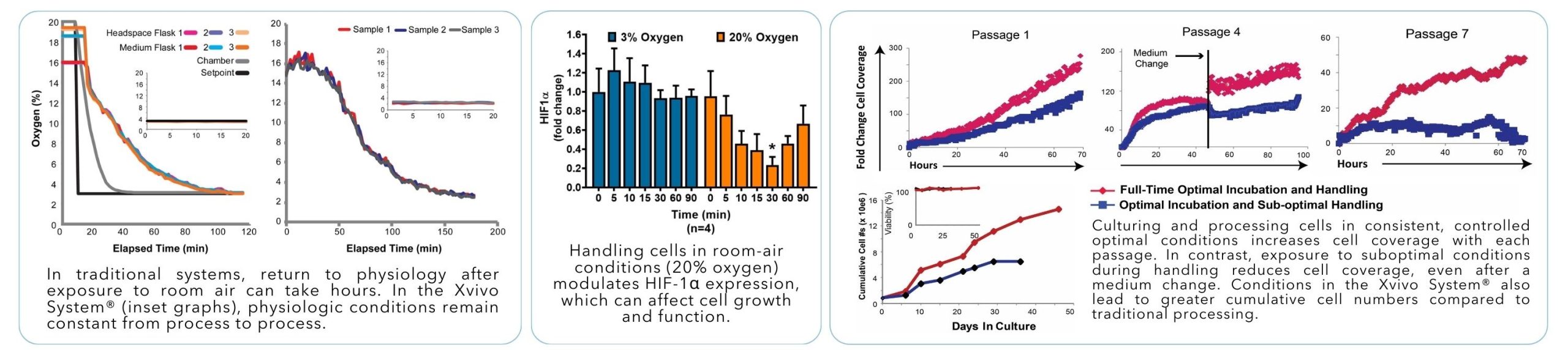
Over 30 years ago, our founder developed the first device capable of precisely controlling oxygen levels within an incubator. This simple but groundbreaking innovation exposed a critical flaw in traditional cell culture methods: existing systems failed to provide cells with physiologically relevant conditions. Recognizing this gap laid the foundation for our Cytocentric® philosophy, where the needs of cells come first.
Below is just some of our data demonstrating why maintaining physiologic oxygen levels and comprehensive environmental control, the unifying goal behind all of our products, produce superior results compared to conventional methods that expose cells to ambient air. Through side-by-side comparisons, key performance metrics, and real-world examples, we highlight the profound impact that true physiological conditions have on cell health, behavior, and therapeutic efficacy.
Our science shows that when cells are given what they truly need, the outcomes aren’t just better – they’re revolutionary.
Commonly asked questions:
Why does exposing my cells to room air for small amounts of time matter?
Cellular responses to oxygen are mediated in part by hypoxia-induced factors (HIF-1 and -2 protein families). These transcription factors control a wide variety of cell functions, and are among the most rapidly regulated proteins found to date.
HIF-1 alpha is downregulated at the protein level within minutes of oxygen elevation, which can in turn affect additional transcription factors and cellular behavior. Eliminating or minimizing pericellular oxygen changes is critical to avoid destabilizing HIF proteins.
What is the oxygen level in the traditional open-room CO₂-controlled incubators I have been using?
Each time the incubator door is opened, CO₂ escapes into the room. When the door is closed, the incubator restores its 5% CO₂ setpoint by injecting CO₂, which necessarily reduces the oxygen percentage inside the chamber. In incubators with an open water pan, water vapor can occupy up to ~8% of the chamber’s gas volume, displacing oxygen even further.
When we measured oxygen levels in standard open-room incubators set to 5% CO₂, we found an average of 17.2 ± 0.3% O₂ after normal door access. Each door opening triggered another cycle of CO₂ loss and reinfusion, driving oxygen levels down again, with smaller incubators showing larger swings in gas levels even during normal use.
For researchers who assume their cultures experience stable atmospheric oxygen, these routine fluctuations may influence cell metabolism, gene expression, and experimental reproducibility.
I have seen researchers use 1%, 2%, 5%, 8%, or 10% oxygen. What oxygen level should I use for my cells?
Oxygen levels in vivo vary widely depending on local physiology. Circulating venous blood is typically around ~5% O₂, while certain tissues experience much lower levels. Bone marrow, stem cell niches, and fetal tissues often exist in relatively low oxygen environments. Poorly vascularized regions, such as cartilage or the interior of solid tumors, can approach near-anoxic conditions.
Because oxygen exposure strongly influences cell metabolism, signaling, and differentiation, the most appropriate oxygen level depends on the native physiology of the cell or tissue you are modeling. We recommend consulting the scientific literature related to your specific model and performing preliminary experiments across a range of oxygen levels to determine optimal conditions for your cells.
What oxygen level does my cell culture experience in the bottom of a cell culture vessel?
In addition to the oxygen concentration set in the incubation chamber, several factors determine the actual oxygen level at the cell layer.
One key factor is medium depth. Deeper media increase the diffusion distance, which can slow equilibration and reduce the oxygen available at the bottom of the vessel.
Culture vessel design also plays a role. Some specialized vessels use oxygen-permeable materials that allow oxygen exchange through the bottom of the plate or flask. In static culture, this can shorten the diffusion path and allow oxygen changes in the chamber to reach the cells more quickly.
Finally, cell density and metabolic activity strongly influence local oxygen levels. As cell numbers increase, oxygen consumption rises, which can significantly lower the pericellular oxygen concentration near the cell layer.
For more consistent and reproducible conditions, maintain controlled chamber oxygen levels, consistent media depth, and similar cell densities between experiments.
What about the oxygen levels in the incubator outside of the C-Chamber?
Escaped gases used to control conditions inside the C-Chamber can escape to the interior of the incubator, which may reduce the partial pressure of oxygen. To prevent pressure buildup during gas infusion, C-Chambers include pressure-equalization features. A soft magnetic door seal allows the door to briefly “burp” and reseal if pressure rises above ambient levels. Additionally, chambers can be designed with an additional hose barb to vent excess gas to the outside of the incubator via tubing. For the most precise environmental control, we recommend a dedicated C-Chamber and controller for each oxygen or gas condition required.
How can I get the best results if I have to handle my cells in a standard room-air Biological Safety Cabinet (BSC)?
If cell handling must be done in room air, such as in a standard biological safety cabinet, the pre-equilibration of cell culture media and other solution can minimize undesirable pericellular oxygen level swings.
What is the longest length of time I can have my cells out in the BSC?
The longer that cell media and other liquid components are mixed with HEPA-filtered room air in the BSC, the more they equilibrate with room air, which is supraphysiologic. Sensitivity to changes in oxygen levels differ based on cell type, though a general rule is that the longer that cells are out of their optimal physiologic range, the more likely they are to be affected by those conditions which, in turn, affects downstream applications.
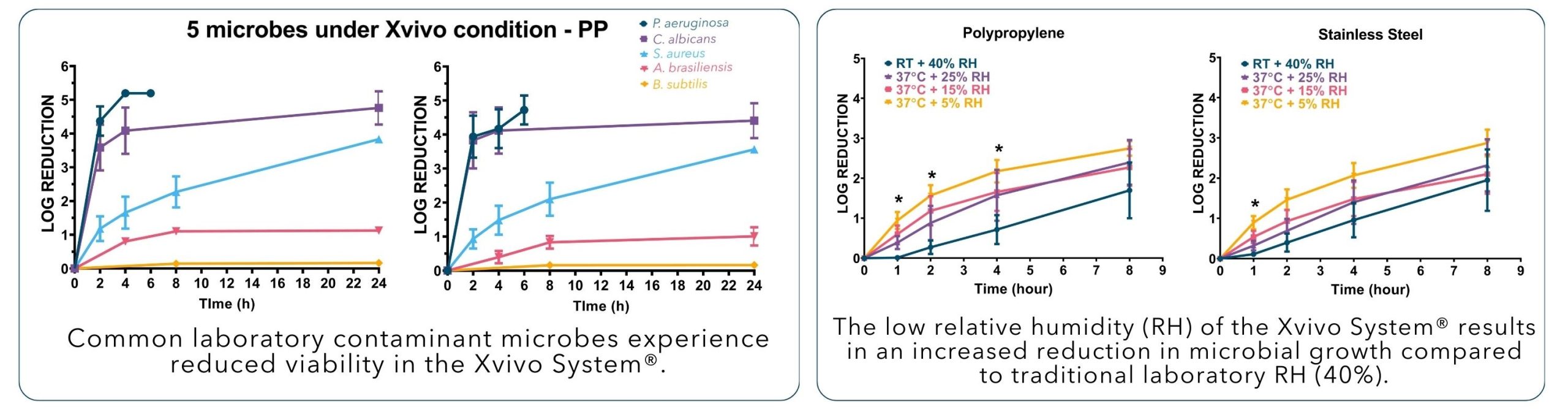
How does the Xvivo System offer cells a better environment than a traditional laboratory or cleanroom setup?
Explore the data below to see how the Xvivo System provides constant, optimal growth conditions and mitigates contamination during cell processing.
The Xvivo System® mitigates contamination due to a patented, self-sanitizing process that reduces unwanted microbial growth*
Cells experience better growth in the Xvivo System® due to consistent and controlled physiologic conditions
The Xvivo System® mitigates edge effect by providing precise and consistent temperature control across all surfaces


Our Scientific Posters
Our Publications
Our customers are the strongest testament to the effectiveness of our Cytocentric® approach. Click below to explore their peer-reviewed scientific articles and see the proof in action.
*This process is protected by US and International Patents including US Patent 11326140